

Press release by Lumosa Therapeutics
LUMOSA THERAPEUTICS ANNOUNCES POSITIVE RESULTS FROM LT3001 PHASE 2A CLINICAL TRIAL IN ACUTE ISCHEMIC STROKE
Lumosa Therapeutics (Lumosa; 6535.TWO) announced today that its Phase 2a clinical trial of LT3001 successfully met its primary safety endpoint. Further, efficacy showed potential for improvements in neurological and functional outcomes based on the majority of LT3001 patients with baseline National Institutes of Health stroke scale (NIHSS) ≥6 demonstrating pronounced neurological improvement (NIHSS improvement ≥4 points). This study was conducted in the U.S. and Taiwan, and was designed to evaluate LT3001 versus placebo/control in subjects with Acute Ischemic Stroke (AIS) within 24 hours after stroke symptoms onset.
“Considering the extensive global unmet medical need for patients and their families battling stroke, we are extremely pleased by the outcome of this study,” said Rongjin Lin, Ph.D., President & CEO, Lumosa Therapeutics.
“LT3001 represents a completely novel drug design in stroke treatment -- combining thrombolytic and neuroprotective properties into a single molecule which may confer unique efficacy and safety properties permitting an extended treatment window,” said Thomas Devlin, MD, PhD, Professor of Neurology, University of Tennessee Health Science Center and principal investigator of the study. “The results from this landmark study pave the way for future studies where LT3001 can be delivered intravenously within a 24-hour time window, either alone or in combination with mechanical stroke treatment. Such studies potentially represent the most impactful clinical studies in the history of stroke treatment"
Lumosa conducted a multicenter, double-blind, single-dose, randomized, and placebo-controlled prospective Phase 2a clinical study in the U.S. and Taiwan. The study enrolled twenty-four subjects and was designed to evaluate LT3001 in AIS patients within 24 hours after stroke symptoms and ineligible to receive IV recombinant tissue-type plasminogen activator (tPA, also known as IV tPA) and/or endovascular thrombectomy. The primary endpoint was the occurrence of symptomatic intracranial hemorrhage (sICH) within 36 hours after dosing. Secondary endpoints include vital measures of efficacy including the mRS (Modified Rankin Score) and change in NIHSS (NIH Stroke Scale).
Twenty-four patients were randomly assigned to receive LT3001 or placebo within 24 hours after onset of ischemic stroke. Participants in the LT3001 and placebo groups were a mean age of 62 (SD±13) and 68 (SD±9) years with median NIHSS of 6 ( Range 4-24) and 5 (Range 4-17), respectively. The median time to treatment was 20 and 18.5 hours for patients who received LT3001 and placebo, respectively. Treatment with LT3001 appeared safe with no evidence of increased risk of sICH. While the sample size was limited, more subjects achieved excellent functional outcome (mRS 0-1) at day 90 and pronounced neurological improvement (NIHSS improvement ≥4 points) at day 30 in the LT3001 group. Among participants with baseline NIHSS ≥6 treated with LT3001, 78% showed pronounced neurological improvement.
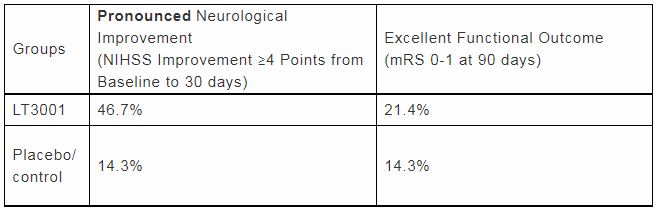
“These data show the potential of LT3001 being administered anywhere in the world, either with or without intervention therapy and the patients that were treated with LT3001 had potential for a significant degree of neurological improvement compared to placebo.” Dr. Devlin added.
In parallel to the successful outcomes of the Phase 2a study, Lumosa is pursuing a two-prong development strategy for LT3001 - In the US, Taiwan and China, Phase 2b multi-dose trials will be conducted in the AIS patients left with no treatment options to expand the potential treatable population. Additionally, a Phase 2 trial will be conducted in AIS patients undergoing endovascular thrombectomy to elucidate outcomes. Lumosa is seeking global partners for later phase development if the arrangement is deemed to accelerate the development program and increase the feasibility of technical success.
According to the World Health Organization (WHO), stroke is the second leading cause of death for people over the age of 60 with approximately 6 million deaths in the world per year. Stroke can be categorized as hemorrhagic (bleeding) or ischemic (lack of blood flow to critical areas of the brain). Studies show ischemic stroke is most common, occurring in about 85% of all stroke cases. Stroke is clearly a disease with significant global unmet medical need. There are 15 million people worldwide who suffer a stroke each year. Although the tPA and surgery are available, 80% of stroke patients are still left with no other treatment options or without desired outcomes.

